CURING LOCALIZED IMMUNE CONDITIONS
Starting with Type 1 Diabetes
At Rezq Bio, we are transforming the treatment of autoimmune and inflammatory diseases, beginning with Type 1 Diabetes (T1D)
With clinically validated in-human safety, our therapies centered on dendritic cells are designed to treat T1D patients at every stage of the disease - with negligible side effects
The focus of our platform is regional inflammation. This localized approach avoids the harsh effects of systemic immune suppression while creating a versatile foundation for treating a spectrum of inflammatory conditions - without significant therapy modifications
T1D IS A global PROBLEM WITH NO SOLUTION
64k
New U.S. Diagnoses Annually
2M
Patients Currently in the U.S.
18M
Patients Worldwide by 2040
$85B
Economic Impact Globally
NO LONGEr A JUVENILE CONDITION
In a 2023 study from Johns Hopkins Bloomberg School of Public Health:
● Up to 62% of T1D cases develop in patients older than 20 (median 24 years)
● 37% of patients were diagnosed after 30 years
LACK OF EFFECTIVE TREATMENTS
There are no FDA-approved therapies for TID proven to…
● Prevent disease onset
● Arrest its progression or
● Restore insulin production safely
T1D Treatment Approaches

platform
Our approach focuses on dendritic cells as they are the regulator cells of the immune system. They can create an inflammatory or tolerant environment
We use our technological platform to convert dendritic cells from a mature (mDC) or inflammatory state, into an immature (iDC) or tolerant state

mDC

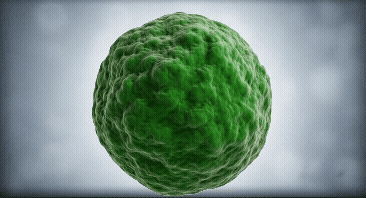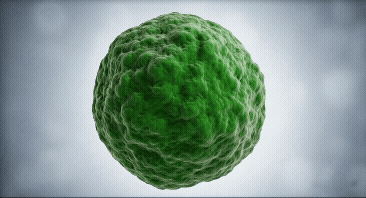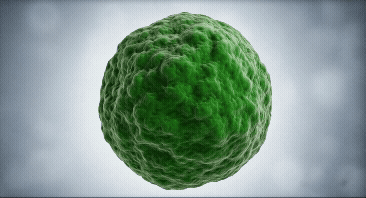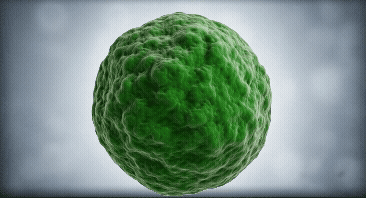
iDC
Our lead P1B/2A ready autologous dendritic cell therapy does this ex-vivo while our microsphere loaded version has performed this preclinically in situ with equally strong performance
Through regional targeting, we can turn off unwanted immune responses locally while not suppressing the entire immune system
Pipeline
Rezq’s pipeline is led by our cell therapy designed to treat early onset T1D. With promising in-human safety it provides a launching point for our multi-disease, dual-application and dual delivery method platform
people
We have built careers in healthcare and are passionate about curing T1D and autoimmune disease
Board and Advisory
We are fortunate to have an accomplished Board and Advisory that believe in our team, science and mission to treat autoimmune disease














